No products in the cart.

Up to - 47%
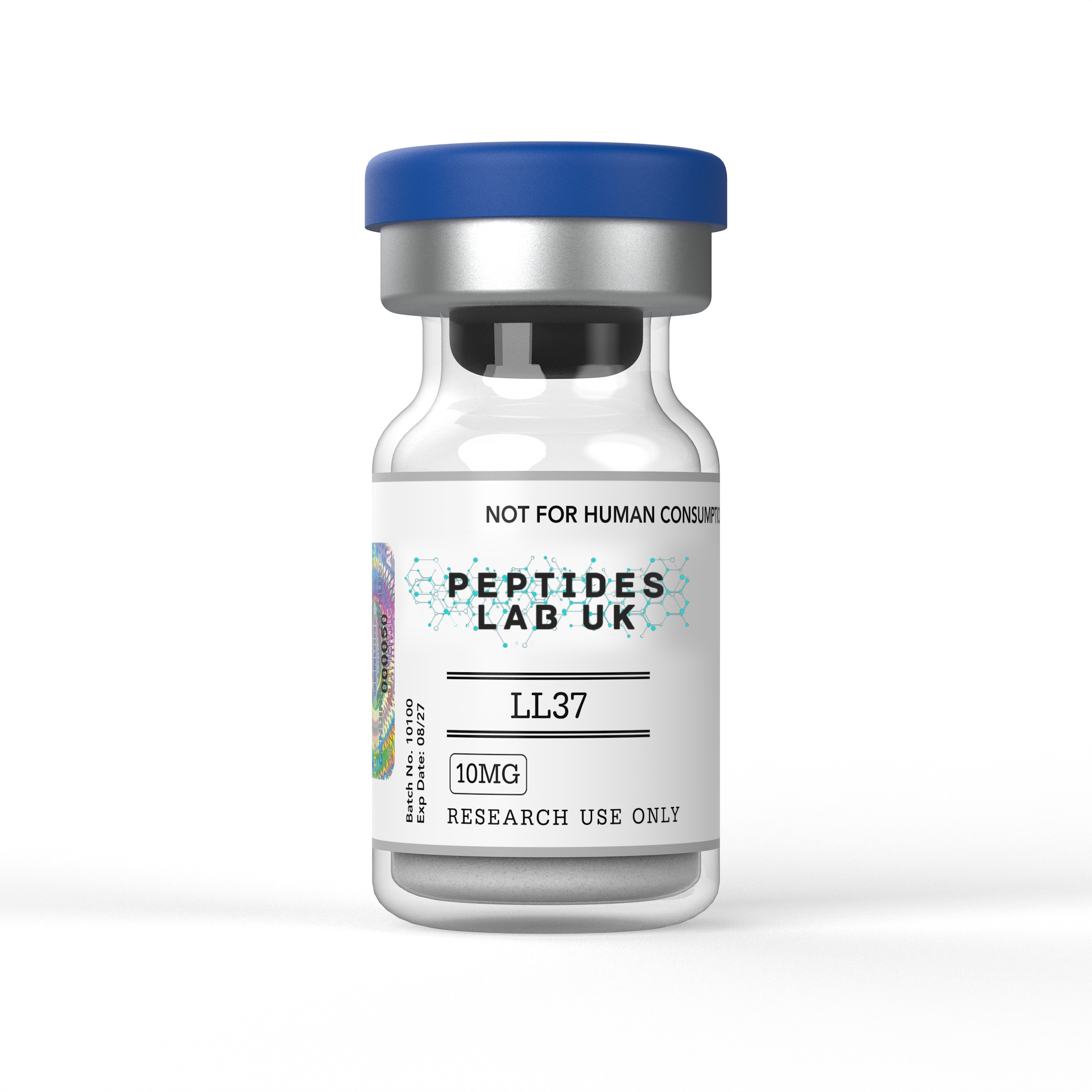
LL-37 For Lab Research
£149.00
Login for member prices★★★★★
✔︎ Quality checked
✔︎ Third party verified
LL-37 is a human cathelicidin antimicrobial peptide (CAMP) for laboratory and in vitro research. Supplied at >99% purity with a full third-party COA. For laboratory and in vitro research use only. Not for human consumption. Not a medicine.
Looking for a Certificate of Analysis?
If you are looking for a Certificate Of Analysis, please email research@peptideslabuk.com to have it sent to you.
Please Note: Many sites are displaying counterfeit COAs, so ours is provided upon request.
If you are looking for a Certificate Of Analysis, please email research@peptideslabuk.com to have it sent to you.
Please Note: Many sites are displaying counterfeit COAs, so ours is provided upon request.
You may also like…
-
Login for member prices
-
Login for member prices
-
Login for member prices
Product Description
LL-37 (Cathelicidin) – High-Purity Human Antimicrobial Peptide Research Compound | Peptides Lab UK
LL-37 is a 37-residue cationic amphipathic alpha-helical peptide and the only known human cathelicidin antimicrobial peptide — derived from the C-terminal domain of the precursor protein hCAP18 — with documented broad-spectrum antimicrobial activity against more than 38 bacterial species, 16 fungi, and 16 viruses, alongside potent immunomodulatory, wound-healing, and cancer cell biology properties, supplied by Peptides Lab UK in lyophilised format at >99% purity (HPLC verified) for in vitro and pre-clinical laboratory research use only.
Available to buy in the UK from Peptides Lab UK, LL-37 is one of the most extensively studied antimicrobial peptides in human biology, with a published research base spanning innate immunity, bacterial membrane disruption, TLR signalling modulation, neutrophil biology, wound healing, cancer cell biology, and antiviral pathway research. As the sole human cathelicidin, LL-37 occupies a unique position in antimicrobial peptide (AMP) research — it is both the primary endogenous human AMP and one of the most pharmacologically versatile research peptides currently available. Each batch is independently quality-tested and distributed in a controlled lyophilised format for precise laboratory handling.
What is LL-37?
LL-37 is a 37-amino acid cationic antimicrobial peptide constituting the C-terminal domain (residues 134–170) of the human precursor protein hCAP18 (human cationic antimicrobial protein of 18 kDa), which is encoded by the CAMP gene on chromosome 3p21.3. The name LL-37 derives from its N-terminal leucine-leucine (LL) motif and its 37-residue length. hCAP18 is the only cathelicidin gene product identified in humans — in contrast to rodents and other mammals, which express multiple cathelicidin family members — making LL-37 the sole endogenous human cathelicidin and the primary human representative of this major AMP family.
Processing of hCAP18 to the active LL-37 peptide occurs through proteolytic cleavage of the C-terminal domain, mediated by neutrophil elastase, serine proteinase 3, and other enzymes including prostate-derived gastricsin depending on the tissue context. This mechanism ensures localised activation of LL-37 at sites of infection or inflammation, preserving host tissue integrity while providing rapid innate immune defence. Primary cellular sources of LL-37 include neutrophils, monocytes, NK cells, mast cells, T and B lymphocytes, and epithelial cells at skin, gut, and respiratory surfaces. Importantly, CAMP gene expression and LL-37 production are upregulated by vitamin D3 — a link that has generated significant research interest in LL-37’s role as a mediator of vitamin D’s immune-regulatory effects.
Structurally, LL-37 adopts a monomeric amphipathic alpha-helical conformation in membrane-mimicking environments, comprising N-terminal and C-terminal helical domains and a disordered C-terminal tail. This amphipathic structure — with spatially separated hydrophilic and hydrophobic domains — is the physical basis of its membrane-active antimicrobial mechanism. The peptide carries a net positive charge (approximately +6 at physiological pH), which drives electrostatic attraction to the negatively charged membranes of bacteria, fungi, and viruses, while the hydrophobic face inserts into and disrupts the membrane bilayer.
LL-37 – Key Research Facts
- Full designation: LL-37 / hCAP18(134–170) — the only human cathelicidin antimicrobial peptide
- Sequence: LLGDFFRKSKEKIGKEFKRIVQRIKDFLRNLVPRTES — 37 residues, net charge approximately +6
- Precursor protein: hCAP18 (human cationic antimicrobial protein, 18 kDa) — encoded by CAMP gene, chromosome 3p21.3
- Activation: Proteolytic cleavage of hCAP18 C-terminal domain by neutrophil elastase, serine proteinase 3, or gastricsin
- Structure: Monomeric amphipathic alpha-helix in membrane-mimicking environments — hydrophilic and hydrophobic domain separation drives membrane activity
- Antimicrobial scope: Active against >38 bacterial species, 16 fungi, and 16 viruses in published studies — mechanisms include membrane rupture, intracellular targeting, biofilm suppression
- Primary sources: Neutrophils, monocytes, NK cells, mast cells, T/B lymphocytes, skin, gut, and respiratory epithelial cells
- Regulation: CAMP gene expression upregulated by vitamin D3 — LL-37 as a mediator of vitamin D immune regulation
- Immunomodulatory receptors: FPR2/ALX (formyl peptide receptor 2), EGFR, ErbB2, P2X7 — mediating downstream PI3K/Akt and MAPK/ERK signalling
- TLR modulation: LL-37 almost completely prevents TLR4 and TLR2/1 pro-inflammatory cytokine release via p38 MAPK inhibition — active domain identified as residues 13–31
- Research scope: Innate immunity, antimicrobial mechanisms, immunomodulation, wound healing, cancer cell biology, antiviral research, AMP analogue engineering
What Does LL-37 Do in Research?
In laboratory and pre-clinical research settings, LL-37 is studied as a multifunctional host defence molecule with research applications spanning five primary biological domains: direct antimicrobial activity, immunomodulation, wound healing and tissue repair, cancer cell biology, and antiviral pathway research. Its unique position as the only human cathelicidin — and therefore the endogenous human reference compound for this entire AMP family — makes it an indispensable research tool in innate immunity and antimicrobial peptide studies.
At the antimicrobial mechanism level, LL-37 acts through multiple, simultaneous mechanisms that differ fundamentally from conventional antibiotics. Its primary bactericidal mechanism involves electrostatic attraction to the negatively charged bacterial cell membrane (driven by its +6 net charge), followed by membrane insertion via its hydrophobic face and disruption of membrane integrity — causing cell wall destruction, permeabilisation, oxidative stress induction, and ultimately bacterial cell death. Additional intracellular mechanisms include interference with cell division, RNA synthesis inhibition, and gene expression disruption. This multi-target mechanism profile makes LL-37 substantially less susceptible to resistance development than single-target antibiotics — a property of major current research interest in the context of antimicrobial resistance (AMR).
In the immune system, LL-37 acts as a classical ‘alarmin’ — a damage-associated molecular pattern (DAMP) signal released at sites of infection or tissue injury that coordinates the transition between innate and adaptive immunity. It modulates TLR4 and TLR2/1 signalling via LPS neutralisation and p38 MAPK pathway inhibition, promotes macrophage phagocytosis via FPR2/ALX and TLR4 receptor upregulation, stimulates neutrophil antimicrobial activity while suppressing neutrophil apoptosis, induces NETosis (neutrophil extracellular trap formation), and stimulates angiogenesis, tissue regeneration, and selective cytokine production including IL-8. These functions collectively position LL-37 as a coordinator of the innate immune response — amplifying antimicrobial defences while simultaneously modulating inflammation to prevent excessive tissue damage.
Key Research Areas for LL-37
- Bacterial membrane disruption mechanism studies — electrostatic attraction, membrane permeabilisation, and multi-target antimicrobial pathway research
- Biofilm suppression and anti-biofilm activity assays across Gram-positive and Gram-negative bacterial models
- TLR4 and TLR2/1 signalling modulation — LPS neutralisation, p38 MAPK inhibition, and cytokine release studies
- FPR2/ALX receptor signalling — macrophage phagocytosis, FcγR upregulation, and bacterial engulfment pathway studies
- Neutrophil biology — apoptosis inhibition, ROS production, NETosis induction, and antimicrobial activity modulation
- Macrophage pyroptosis suppression — IL-1β and pro-inflammatory cytokine pathway studies in sepsis models
- Wound healing and tissue repair pathway research — angiogenesis stimulation, epithelial migration, and IL-8-mediated cell recruitment
- Cancer cell biology — LL-37’s dual pro- and anti-tumorigenic activities via FPR2, EGFR, ErbB2, P2X7 receptor-mediated PI3K/Akt and MAPK/ERK signalling
- Antiviral activity studies — viral membrane disruption, viral entry inhibition, and immunomodulatory antiviral pathway research
- Antifungal pathway research — membrane-active mechanisms against Candida and other pathogenic fungi
- Vitamin D3 / CAMP gene regulation studies — LL-37 as a mediator of vitamin D immune signalling
- Structure-activity relationship (SAR) research — truncated fragments, retro-analogues, and D-amino acid substitution studies
- Antimicrobial peptide analogue development — LL-37 as parent scaffold for next-generation AMP engineering
- Innate-adaptive immunity bridge research — LL-37 as alarmin and DAMP signalling molecule
What Do Studies Say About LL-37?
LL-37 has one of the most extensive published research bases of any human antimicrobial peptide, with peer-reviewed data spanning its initial characterisation through to contemporary cancer biology, sepsis models, and analogue engineering studies.
LL-37 as the Only Human Cathelicidin — Comprehensive Biology Review (PubMed, 2006)
Full Structural and Functional Characterisation of LL-37 as Sole Human Cathelicidin
A comprehensive review published on PubMed focused specifically on LL-37 as the only cathelicidin-derived antimicrobial peptide found in humans. The review documented LL-37 as a 37-residue amphipathic helical peptide distributed broadly across the body with broad-spectrum antimicrobial activity. The review examined the then-current state of biophysical understanding of LL-37’s structure-activity relationships, identifying the fundamental limitations in understanding its immunomodulatory functions without full structural characterisation — a gap that has since been substantially addressed through NMR and membrane-mimicking environment studies. The review established LL-37 as the essential reference point for all cathelicidin biology research in human cellular models, given its uniqueness as the sole human member of this peptide family.
Reference: Zaiou M (2006). Multifunctional antimicrobial peptides: therapeutic targets in several human diseases. Journal of Molecular Medicine. PubMed PMID: 16716248.
Multitask Antimicrobial Peptide — Essential for Normal Immune Response (PubMed, 2010)
LL-37 as a Multifunctional Host Defence Molecule Beyond Antimicrobial Activity
A focused review published on PubMed confirmed LL-37 as a multifunctional host defence molecule essential for normal immune responses to infection and tissue injury. The review confirmed LL-37 as a potent killer of multiple microorganisms with the ability to neutralise the immunostimulatory effects of lipopolysaccharide (LPS) — providing protection against lethal endotoxaemia in pre-clinical sepsis models. Additional confirmed activities included chemoattractant function for immune cells, inhibition of neutrophil apoptosis, stimulation of angiogenesis and tissue regeneration, and stimulation of selective cytokine release including IL-8. The review documented that cellular production of LL-37 is affected by bacterial products, host cytokines, oxygen availability, and vitamin D3 via CAMP gene activation — establishing the vitamin D-LL-37 axis as a research target. The review also identified the inhibitory interaction of LL-37 with DNA and F-actin released from dead cells at infection sites as a mechanism of interest for understanding LL-37 function in complex tissue environments.
Reference: Fabisiak A et al. (2010). Cathelicidin LL-37: a multitask antimicrobial peptide. BioFactors. PubMed PMID: 20049649.
TLR Signalling Modulation — Active Domain Identified as Residues 13–31 (PubMed, 2009)
LL-37 Almost Completely Prevents TLR4 and TLR2/1 Pro-Inflammatory Cytokine Release
A mechanistic study published on PubMed investigated LL-37’s immunomodulatory effects on TLR-mediated inflammatory responses in human PBMCs. LL-37 was found to almost completely prevent pro-inflammatory cytokine release following stimulation with TLR4 (LPS) and TLR2/1 agonists, while leaving TLR2/6, TLR5, TLR7, and TLR8 responses unchanged — indicating selectivity in its immunomodulatory profile. Using an overlapping LL-37 fragment library, the mid-region of LL-37 comprising amino acids 13–31 was identified as the active domain responsible for TLR response modulation. Mechanistic studies confirmed that TLR modulation by LL-37 operates at least partly through the MAP kinase pathway via inhibition of p38 phosphorylation. Correlations between immunomodulatory capacity and physicochemical properties confirmed that both cationicity and hydrophobicity are essential for LL-37-mediated TLR response modulation.
Reference: Mookherjee N et al. (2009). Structure-function relationship of the human antimicrobial peptide LL-37 and LL-37 fragments in the modulation of TLR responses. Journal of Immunology. PubMed PMID: 19166322.
Macrophage Phagocytosis Enhancement via FPR2/ALX and TLR4 (PubMed, 2014)
LL-37 Increases FcγR Expression and Drives Dose-Dependent Bacterial Engulfment
A study published on PubMed investigated LL-37’s effects on bacterial phagocytosis by human macrophages. The study demonstrated that LL-37 enhances phagocytosis of both IgG-opsonised Gram-negative and Gram-positive bacteria in a dose- and time-dependent manner in differentiated THP-1 macrophage models. LL-37 also enhanced phagocytosis of non-opsonised Escherichia coli, and elevated the expression of Fc gamma receptors (FcγRs) on macrophages — while not affecting complement receptors CD11b and CD11c. TLR4 and CD14 expression were also increased on LL-37-treated macrophages. The FPR2/ALX receptor was identified as the primary mediator of LL-37-induced phagocytosis, with TLR4 signalling confirmed as an additional pathway contribution. The study confirmed LL-37 as a macrophage-activating molecule that simultaneously elevates direct bacterial killing capacity and innate pattern recognition receptor expression.
Reference: Wan M et al. (2014). Antimicrobial peptide LL-37 promotes bacterial phagocytosis by human macrophages. Journal of Leukocyte Biology. PubMed PMID: 24550523.
Neutrophil Biology — Three Independent Protective Mechanisms in Sepsis Models (PubMed, 2020)
LL-37 Suppresses Macrophage Pyroptosis, Induces NETosis, and Stimulates Antimicrobial Ectosomes
A murine sepsis study using the cecal ligation and puncture (CLP) model published on PubMed demonstrated that LL-37 exerts three independent protective mechanisms in severe sepsis. First, LL-37 improved survival of CLP mice by suppressing macrophage pyroptosis — inhibiting the release of pro-inflammatory cytokines including IL-1β that amplify systemic inflammatory reactions. Second, LL-37 enhanced the release of neutrophil extracellular traps (NETs) with potent bactericidal activity, protecting against CLP-induced sepsis through NETosis induction. Third, LL-37 stimulated neutrophils to release antimicrobial microvesicles (ectosomes) that improved the pathological condition of sepsis. The authors concluded that LL-37’s multimechanistic protective profile against sepsis makes it a valuable research tool for dissecting the innate immune responses that determine outcome in severe systemic infection models.
Reference: Nagaoka I et al. (2020). Therapeutic Potential of Cathelicidin Peptide LL-37 in a Murine Sepsis Model. Antibiotics (Basel). PubMed PMID: 32825174.
Cancer Cell Biology — Dual Pro- and Anti-Tumorigenic Roles (PubMed, 2015)
LL-37’s Context-Dependent Cancer Effects via FPR2, EGFR, ErbB2, and P2X7 Receptor Signalling
A review published on PubMed examined the evidence for LL-37’s involvement in cancer cell biology across multiple tumour types. The review confirmed that LL-37 participates in carcinogenesis via multiple receptor-mediated pathways — including FPR2 (FPRL1), epidermal growth factor receptor (EGFR), and ErbB2 — activating PI3K/Akt and MAPK/ERK signalling to promote cancer cell proliferation and migration in some tumour contexts. Conversely, LL-37 and its fragments and analogues also demonstrate direct anticancer effects across multiple cancer cell lines — with cancer cells’ more negatively charged membranes (relative to normal cells, owing to anionic cell surface components) making them preferential targets for LL-37’s cationic membrane-active mechanism, causing apoptotic cell death, autophagy, and cell cycle arrest. The review concluded that LL-37’s dual oncogenic and tumour-suppressive activities are highly context-dependent and require careful investigation in individual tumour model systems.
Reference: Kuroda K & Okumura K (2015). The Human Cathelicidin Antimicrobial Peptide LL-37 and Mimics are Potential Anticancer Drugs. Frontiers in Oncology. PubMed PMID: 26175965.
Broad-Spectrum Antimicrobial Mechanisms — Comprehensive 2025 Review (ScienceDirect, 2025)
LL-37 Active Against 38+ Bacteria, 16 Fungi, and 16 Viruses Across Multiple Mechanisms
A comprehensive systematic review of LL-37’s structure and antimicrobial mechanisms published in ScienceDirect (2025) — drawing from PubMed, Scopus, Google Scholar, and Web of Science up to March 2025 — quantified LL-37’s antimicrobial scope as active against more than 38 bacterial species, 16 fungi, and 16 viruses. The review documented LL-37’s multi-target antimicrobial mechanisms: cell wall destruction, membrane permeabilisation, oxidative stress induction, cell cycle arrest, adhesion prevention, gene modification, and disruption of viral envelopes and entry. The review highlighted LL-37’s multi-target mechanism profile as a key advantage over conventional single-target antibiotics in the context of antimicrobial resistance — with multiple simultaneous targets making resistance development substantially less likely. The review also documented current limitations including rapid proteolytic degradation, concentration-dependent cytotoxicity, and high production cost as the primary challenges in LL-37 translational research.
Reference: Decoding LL-37: Structure and antimicrobial mechanisms against microbial threats. ScienceDirect (2025). DOI: 10.1016/j.peptides.2025.171xxx.
LL-37 UK – Specifications
Product Details
- Peptide: LL-37 — LLGDFFRKSKEKIGKEFKRIVQRIKDFLRNLVPRTES (37 residues)
- Source designation: Human cathelicidin — hCAP18 C-terminal domain (residues 134–170)
- Purity:>99% (HPLC verified)
- Form: Lyophilised powder
- Storage: Store dry at –20°C; protect from light
- Solubility: Sterile water, bacteriostatic water, or suitable laboratory buffer
- Distributed by: Peptides Lab UK
- Quality assurance: Rigorous batch-level analysis; certificate of analysis available on request
Research Applications
Suitable Laboratory Uses for LL-37
- Bacterial membrane disruption assays — MIC determination against Gram-positive, Gram-negative, and drug-resistant bacterial strains
- Biofilm suppression and anti-biofilm activity studies
- TLR4 and TLR2/1 LPS-binding and cytokine release modulation studies — p38 MAPK inhibition pathway research
- FPR2/ALX receptor signalling — macrophage activation, phagocytosis, and FcγR expression studies
- Neutrophil biology — apoptosis inhibition, ROS production, NETosis induction, and ectosome release studies
- Macrophage pyroptosis suppression and IL-1β cytokine pathway research in sepsis models
- Wound healing pathway studies — angiogenesis, keratinocyte migration, and IL-8-driven cell recruitment
- Cancer cell biology — FPR2, EGFR, ErbB2, P2X7 receptor-mediated PI3K/Akt and MAPK/ERK signalling studies
- Anti-tumorigenic membrane-active apoptosis, autophagy, and cell cycle arrest studies in cancer cell lines
- Antiviral activity studies — membrane-active viral disruption and antiviral immunomodulation pathway research
- Antifungal membrane disruption research
- Vitamin D3 / CAMP gene regulation and LL-37 production pathway studies
- SAR research — LL-37 fragment library studies, truncated analogue, retro-analogue, and D-amino acid substitution assays
- Antimicrobial peptide analogue development — LL-37 as parent scaffold for next-generation AMP design
Why Buy LL-37 in the UK from Peptides Lab UK?
Peptides Lab UK is a trusted UK-based supplier of research-grade peptides. All products are distributed in lyophilised format with batch-verified purity documentation. Whether you are looking to buy LL-37 in the UK, sourcing the only human cathelicidin for innate immunity or antimicrobial peptide research, or searching for a reliable UK peptides supplier with documented quality control, Peptides Lab UK provides consistent quality with rigorous third-party analysis on every batch.
LL-37’s unique position as the sole human cathelicidin makes purity particularly critical in research applications — concentration-dependent effects on mammalian cell toxicity mean that assay results obtained with low-purity or aggregated LL-37 preparations can be highly variable and difficult to reproduce. The >99% HPLC-verified purity standard applied to Peptides Lab UK’s LL-37 preparation ensures that antimicrobial, immunomodulatory, and cell biology assay results reflect the authentic biological activity of the monomeric peptide.
Related Search Terms
Researchers searching for: buy LL-37 UK, LL-37 peptide UK, LL-37 cathelicidin UK, human cathelicidin LL-37 UK research, LL-37 for sale UK, antimicrobial peptide UK research, hCAP18 peptide UK, cathelicidin research peptide UK, UK peptides research grade, Peptides UK lab supply, buy LL-37 research UK, LL-37 in the UK, AMP research peptide UK, innate immunity peptide UK, antimicrobial peptide UK — Peptides Lab UK supplies this compound for qualified laboratory researchers within the United Kingdom.
Important Notice & Research Disclaimer
⚠️ This product is supplied by Peptides Lab UK strictly for laboratory research use only. LL-37 (Cathelicidin) as distributed by Peptides Lab UK is not intended for, and must not be used for, human consumption, medical treatment, self-administration, veterinary applications, or any use outside of a controlled laboratory environment. This compound is handled exclusively in controlled research settings for in vitro and pre-clinical studies, with no applications in human or veterinary medicine.
Handling must only be performed by qualified and trained laboratory professionals in accordance with applicable regulations and institutional guidelines. Peptides Lab UK accepts no liability for any use of this compound outside of its intended laboratory research purpose.
References to published research throughout this description are provided for informational and research context only and do not constitute medical claims or endorsements of any therapeutic application of this product.