No products in the cart.

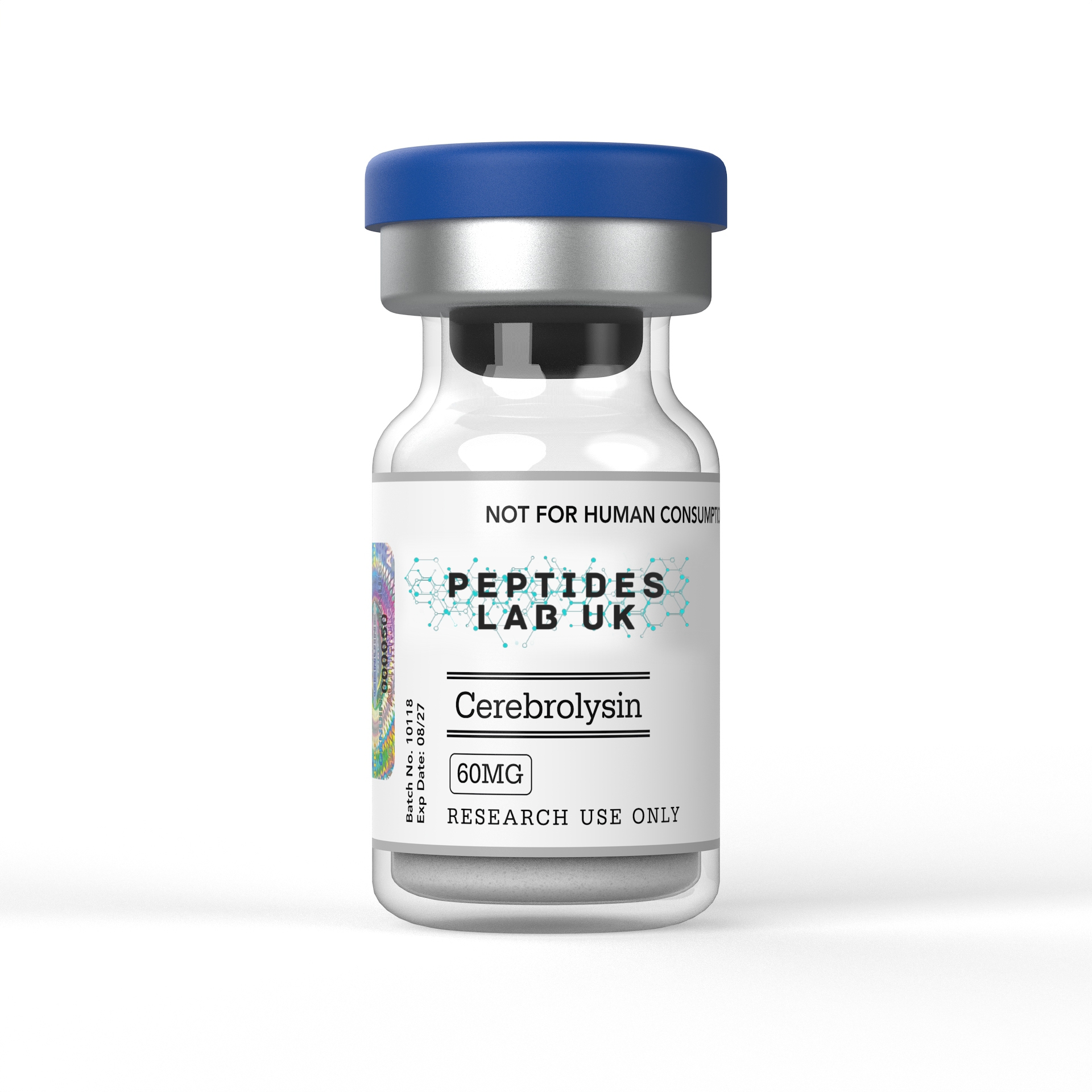
Buy Cerebrolysin UK For Lab Research
£130.00
Login for member prices★★★★★
✔︎ Quality checked
✔︎ Third party verified
Cerebrolysin is a neuropeptide mixture from purified porcine brain proteins, used in laboratory and in vitro research into neuroprotection. For laboratory and in vitro research use only. Not for human consumption. Not a medicine.
Looking for a Certificate of Analysis?
If you are looking for a Certificate Of Analysis, please email research@peptideslabuk.com to have it sent to you.
Please Note: Many sites are displaying counterfeit COAs, so ours is provided upon request.
If you are looking for a Certificate Of Analysis, please email research@peptideslabuk.com to have it sent to you.
Please Note: Many sites are displaying counterfeit COAs, so ours is provided upon request.
You may also like…
-
Login for member prices
-
Login for member prices
-
Login for member prices
Product Description
Cerebrolysin – High-Purity Neuropeptide Research Preparation | Peptides Lab UK
Cerebrolysin is a standardised neuropeptide preparation derived from purified porcine brain proteins, composed of low-molecular-weight peptides and free amino acids with documented neurotrophic-like activity, supplied by Peptides Lab UK in lyophilised format at >99% purity (HPLC verified) for in vitro and pre-clinical laboratory research use only.
Available to buy in the UK from Peptides Lab UK, Cerebrolysin is one of the most extensively researched neuropeptide preparations in the field of neuroscience, with a published literature base spanning pre-clinical models of neurodegeneration, brain injury, and cognitive pathway research, as well as multiple randomised controlled clinical trials. Each batch is independently quality-tested and distributed in a controlled lyophilised powder form, suitable for precise laboratory handling and storage.
What is Cerebrolysin?
Cerebrolysin is a parenterally administered neuropeptide preparation consisting of a standardised mixture of low-molecular-weight peptides (approximately 25% by weight) and free amino acids (approximately 75% by weight), produced through a controlled enzymatic breakdown of purified porcine brain proteins. The peptide fraction — defined as peptides with a molecular weight below 10 kDa — is considered the biologically active component, with published research characterising its neurotrophic-like properties as mimicking the actions of endogenous neurotrophic factors including BDNF (brain-derived neurotrophic factor), NGF (nerve growth factor), and CNTF (ciliary neurotrophic factor).
Originally developed in Austria, Cerebrolysin has been the subject of more than five decades of pre-clinical and clinical investigation. It is approved for treatment of stroke in several European and Asian countries and is authorised in Austria, making it one of the few neuropeptide preparations with a documented regulatory history. However, as supplied by Peptides Lab UK, this product is strictly intended for laboratory research use only and has no approved medical or therapeutic application in this context.
A distinguishing feature of Cerebrolysin in the published literature is its multi-target mechanism — unlike single-molecule compounds, Cerebrolysin’s peptide mixture acts simultaneously on multiple neurotrophic and neuroprotective pathways, a profile that has led researchers to describe it as a ‘pleiotropic’ or ‘multi-target’ neuropeptide preparation in pre-clinical and clinical review literature.
Cerebrolysin – Key Research Facts
- Composition: ~25% low-molecular-weight peptides (<10 kDa) and ~75% free amino acids
- Source: Enzymatic hydrolysate of standardised purified porcine brain proteins
- Mechanism: Neurotrophic-like activity; mimics BDNF, NGF, and CNTF in experimental models
- Mode: Pleiotropic, multi-target neuropeptide preparation
- Research areas: Neuroprotection, neuroplasticity, neurogenesis, and cognitive pathway studies
- Approved use: Treatment of stroke (Austria and select other countries) — not applicable to Peptides Lab UK supply
- Key signalling pathway: Activation of PI3K/Akt/GSK-3β intracellular signalling in Alzheimer’s-related research
- Comparator studies: Published evidence demonstrating distinct peptide composition versus generic alternatives
What Does Cerebrolysin Do in Research?
In laboratory and pre-clinical research settings, Cerebrolysin is investigated for its effects on a wide range of neurotrophic and neuroprotective signalling pathways. Its multi-target profile has made it the subject of studies spanning neurodegeneration models, brain injury research applications, cognitive function, and neural stem cell research.
Pre-clinical studies have demonstrated that Cerebrolysin can cross the blood-brain barrier, a property that distinguishes it from many larger neuropeptide preparations and expands its research utility in in vivo models. Documented pre-clinical effects include reductions in β-amyloid accumulation and tau-related pathology in Alzheimer’s disease models, attenuation of neuroinflammation and oxidative stress, modulation of excitotoxic signalling, promotion of neurogenesis, and support for neuroplasticity and synaptic repair.
A 2024 comparator study published on PubMed confirmed that the peptide composition and neurotrophic activity of Cerebrolysin is distinct from several generic preparations marketed as equivalent, with generic alternatives found to lack relevant biological activity in standardised neurotrophic assays — underscoring the importance of batch-verified, high-purity sourcing for reliable research outcomes.
Key Research Areas for Cerebrolysin
- Neurotrophic factor signalling pathway studies (BDNF, NGF, CNTF mimicry)
- Neuroprotection and neurodegeneration pathway research in vitro
- β-amyloid and tau pathology modulation in Alzheimer’s-related experimental models
- PI3K/Akt/GSK-3β intracellular signalling cascade investigations
- Neuroinflammation, oxidative stress, and excitotoxicity pathway studies
- Neuroplasticity, neurogenesis, and synaptic repair research
- Neural stem cell survival and grafting studies in pre-clinical models
- Comparative neuropeptide composition and biological activity analysis
- Structure–activity investigations across low-molecular-weight brain peptide fractions
What Do Studies Say About Cerebrolysin?
Cerebrolysin has one of the most substantial published evidence bases of any research neuropeptide preparation, with peer-reviewed data from pre-clinical models, randomised controlled trials, meta-analyses, and systematic reviews across multiple neurological research areas.
Meta-Analysis: Stroke Recovery – Nine RCTs (Neurological Sciences, 2018)
A pre-planned meta-analysis published in Neurological Sciences combined results from nine prospective, randomised, double-blind, placebo-controlled trials assessing Cerebrolysin’s effects on global neurological improvement during early post-stroke research applications. The combined analysis across 1,879 patients demonstrated superiority of Cerebrolysin over placebo on NIHSS neurological deficit scores at day 30 or 21 (Mann-Whitney effect size 0.60, p<0.0001). The combined number needed to treat for clinically relevant NIHSS changes was 7.7 (95% CI 5.2–15.0). Full-scale ordinal analysis of modified Rankin Scale at day 90 in moderate-to-severe patients also reached statistical significance in favour of Cerebrolysin (MW 0.61, p=0.0118). All nine leave-one-out sensitivity analyses remained statistically significant, confirming robustness of the combined result.
Reference: Muresanu DF et al. (2018). Safety and efficacy of Cerebrolysin in early post-stroke research applications: a meta-analysis of nine randomized clinical trials. Neurological Sciences. DOI: 10.1007/s10072-017-3214-0. PubMed PMID: 29248999.
CARS Meta-Analysis: Motor Recovery After Stroke (PubMed, 2017)
A pre-planned meta-analysis of two identical randomised, double-blind, placebo-controlled trials (CARS-1 and CARS-2) evaluated Cerebrolysin’s effects on motor function research applications during early rehabilitation after acute ischemic stroke. Both studies used 30 ml Cerebrolysin once daily for three weeks, initiated within 24–72 hours of stroke onset, alongside standardised rehabilitation. The combined ARAT (Action Research Arm Test) score at day 90 showed superiority of Cerebrolysin over placebo (MW 0.62, p<0.0001). Early benefit was also confirmed at days 14 and 21 on the NIHSS (MW 0.59, p=0.002), with a number needed to treat of 7.1 (95% CI: 4–22). Safety profile was comparable to placebo across both studies.
Reference: Guekht A et al. (2017). Safety and efficacy of Cerebrolysin in motor function research applications after stroke: a meta-analysis of the CARS trials. Neurological Sciences. DOI: 10.1007/s10072-017-3037-z. PubMed PMID: 28707130.
Alzheimer’s Disease Research Review (PubMed, 2012)
A comprehensive review published on PubMed evaluated Cerebrolysin’s pre-clinical and clinical evidence base in Alzheimer’s disease research. Pre-clinical data demonstrated positive effects on β-amyloid and tau-related pathologies, neuroinflammation, neurotrophic factors, oxidative stress, excitotoxicity, neurotransmission, brain metabolism, neuroplasticity, neuronal apoptosis, neurogenesis, and cognition in experimental models. The review described these as consistent with a neurotrophic-like mode of action, and identified the PI3K/Akt/GSK-3β intracellular signalling pathway as a key mechanism in Cerebrolysin’s experimental effects in Alzheimer’s-related models. Multiple randomised, double-blind clinical trials were cited as showing consistent benefits on global clinical function and cognition in patients with mild to moderate Alzheimer’s disease.
Reference: Alvarez XA et al. (2011). Cerebrolysin in Alzheimer’s disease. Drugs of Today. DOI: 10.1358/dot.2011.47.7.1611637. PubMed PMID: 22013558.
Alzheimer’s Disease Dose-Finding Trial (PubMed, 2010)
A randomised, double-blind, placebo-controlled dose-finding trial published on PubMed evaluated Cerebrolysin at doses of 10, 30, and 60 ml in patients with moderate to moderately severe Alzheimer’s disease (MMSE: 14–20). At week 24, Cerebrolysin improved global clinical function significantly at all three dosages, with dose-specific improvements also observed in cognition, initiation of activities of daily living, and neuropsychiatric symptoms. The preparation was reported as safe and well tolerated across all dosing groups. The authors concluded the results demonstrated efficacy in moderate to moderately severe Alzheimer’s disease, with dose-specific effects consistent with those reported in milder disease.
Reference: Gauthier S et al. (2010). Efficacy and safety of Cerebrolysin in moderate to moderately severe Alzheimer’s disease: results of a randomized, double-blind, controlled trial investigating three dosages of Cerebrolysin. Current Alzheimer Research. PubMed PMID: 20500802.
Neural Stem Cell Survival Study – Alzheimer’s Model (PubMed, 2015)
A pre-clinical study investigated Cerebrolysin as an adjunct to neural stem cell (NSC) therapy in an APP transgenic mouse model of Alzheimer’s disease. Murine NSCs were grafted into the hippocampus, with animals treated systemically with Cerebrolysin and analysed at 1, 3, 6, and 9 months post-grafting. The study found that Cerebrolysin improved the survival of grafted neural stem cells in the transgenic model — an effect not observed without the neuropeptide treatment. The authors concluded that Cerebrolysin’s neurotrophic-like properties may support the viability of stem cell grafts in neurodegeneration research models, with potential relevance for combination research approaches.
Reference: Masliah E et al. (2015). Neuro-peptide treatment with Cerebrolysin improves the survival of neural stem cell grafts in an APP transgenic model of Alzheimer disease. Journal of Alzheimer’s Disease. DOI: 10.3233/JAD-150024. PubMed PMID: 26209890.
Peptide Composition & Biological Activity Comparator Study (PubMed, 2024)
A 2024 study published on PubMed compared the peptide composition and neurotrophic biological activity of Cerebrolysin against several preparations marketed as equivalent generics. The study demonstrated that generic alternatives lacked relevant biological activity in standardised neurotrophic assays, and that their peptide compositions differed significantly from that of Cerebrolysin. The authors concluded that not all neuropeptide preparations are interchangeable for research purposes, highlighting the importance of confirmed peptide composition and verified neurotrophic activity in research-grade sourcing.
Reference: PubMed (2024). Comparing the biological activity and composition of Cerebrolysin with other peptide preparations. PubMed PMID: 38737662.
Cerebrolysin UK – Specifications
Product Details
- Purity:>99% (HPLC verified)
- Form: Lyophilised powder
- Storage: Store dry at –20°C; protect from light
- Solubility: Bacteriostatic water, sterile water, or suitable laboratory solvents
- Distributed by: Peptides Lab UK
- Quality assurance: Rigorous batch-level analysis; certificate of analysis available on request
Research Applications
Suitable Laboratory Uses for Cerebrolysin
- Neurotrophic factor signalling pathway and receptor studies (BDNF, NGF, CNTF mimicry)
- Neuroprotection and neurodegeneration pathway research in vitro
- β-amyloid aggregation, tau pathology, and Alzheimer’s-related pathway studies
- PI3K/Akt/GSK-3β intracellular signalling cascade investigations
- Neuroinflammation and oxidative stress pathway research
- Neuroplasticity, neurogenesis, and synaptic signalling studies
- Neural stem cell viability and survival assays in pre-clinical models
- Neuropeptide composition and biological activity characterisation studies
- Molecular analysis and controlled laboratory experiments
Why Buy Cerebrolysin in the UK from Peptides Lab UK?
Peptides Lab UK is a trusted UK-based supplier of research-grade neuropeptides and peptide preparations. All products are distributed in lyophilised format with batch-verified purity documentation. Whether you are looking to buy Cerebrolysin in the UK, sourcing a research-grade neuropeptide preparation for laboratory use, or searching for a reliable UK peptides supplier with documented quality control, Peptides Lab UK provides consistent quality with rigorous third-party analysis on every batch.
Given the published evidence that generic Cerebrolysin alternatives show significantly different peptide composition and lack comparable biological activity, purity-verified sourcing from a reputable UK peptides supplier is particularly important for consistent, reproducible research outcomes.
Related Search Terms
Researchers searching for: buy Cerebrolysin UK, Cerebrolysin peptide UK, Cerebrolysin for sale UK, neuropeptide preparation UK research, UK peptides research grade, Peptides UK lab supply, neurotrophic peptide UK, buy neuropeptide UK, Cerebrolysin in the UK, BDNF mimetic peptide UK, brain peptide research UK, Cerebrolysin research peptide UK — Peptides Lab UK supplies this compound for qualified laboratory researchers within the United Kingdom.
Important Notice & Research Disclaimer
⚠️ This product is supplied by Peptides Lab UK strictly for laboratory research use only. Cerebrolysin as distributed by Peptides Lab UK is not intended for, and must not be used for, human consumption, medical treatment, self-administration, veterinary applications, or any use outside of a controlled laboratory environment. This compound is handled exclusively in controlled research settings for in vitro and pre-clinical studies.
Handling must only be performed by qualified and trained laboratory professionals in accordance with applicable regulations and institutional guidelines. Peptides Lab UK accepts no liability for any use of this compound outside of its intended laboratory research purpose.
References to clinical trials and published research throughout this description are provided for informational and research context only and do not constitute medical claims or endorsements of any therapeutic application of this product.